|
11/9/2023 0 Comments Electrolytic cell cathode charge
The secondary battery industry has experienced rapid growth, owing primarily to rising sales of wireless devices and cordless tools that cannot be economically powered by primary batteries and come with integral rechargeable batteries. The primary battery is considered a wasteful, environmentally unfriendly technology because of its large pollutant concentration relative to its little energy content. A battery's manufacturing energy is around 50 times that of the energy it contains (a better source is required). They are classified as such by most municipalities, and they must be disposed of separately. Electrolysis, which comes from the Greek word lysis, which meaning "to break down," is a common method for decomposing chemical molecules.īatteries are hazardous garbage because they contain poisonous heavy metals and powerful acids or alkalis. One or more cells are linked in series, parallel, or series-and-parallel arrangements to form a battery.Īn electrolytic cell is an electrochemical cell that drives a non-spontaneous redox reaction with electrical energy. A common example of a galvanic cell is a 1.5 volt cell for consumer use. Electrochemical cells that generate an electric current are known as voltaic or galvanic cells, whereas electrolytic cells generate chemical reactions, such as electrolysis (a more reliable source is needed). Every year, some 15 billion primary batteries are discarded worldwide, with the vast majority of them ending up in landfills.Īn electrochemical cell is a device that can either generate or use electrical energy from chemical reactions. 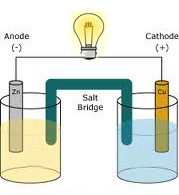
Primary batteries account for over 90% of the $50 billion battery business, while secondary batteries are gaining ground. Small household goods such as flashlights and portable radios are powered by primary cells, which are available in a variety of standard sizes. In a secondary cell, on the other hand, the reaction can be reversed by charging it with a current from a battery charger, renewing the chemical reactants. The electrochemical reaction in the cell is generally irreversible, making the cell unrechargeable.Ĭhemical reactions in the battery deplete the chemicals that generate power when a primary cell is used once these chemicals are depleted, the battery ceases to produce electricity and becomes worthless. A primary cell is a galvanic battery that is meant to be used once and then discarded, as opposed to a secondary cell (rechargeable battery) that may be recharged and reused using electricity. The term "battery" has come to refer to a single galvanic cell, whereas a battery is actually made up of several cells. The voltaic pile, the first electrical battery, was invented by Volta. It usually consists of a salt bridge connecting two distinct metals, or individual half-cells separated by a porous membrane. It is named after Luigi Galvani or Alessandro Volta, respectively. The electrical energy can cause a chemical reaction that would not happen naturally otherwise.Ī galvanic cell, also known as a voltaic cell, is an electrochemical cell that derives electrical energy through spontaneous redox processes within the cell. voltage) of the correct polarity and adequate magnitude. 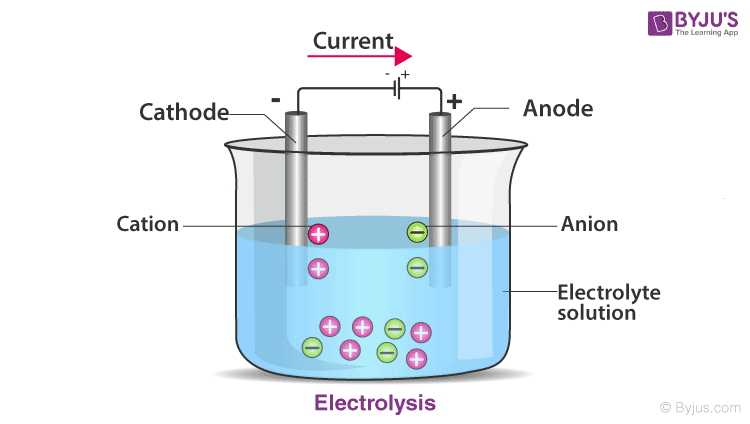
An electrolytic cell can only breakdown a typically stable or inert chemical substance in solution with an external electrical potential (i.e. The ions in the electrolyte are attracted to an electrode with the opposite charge when an external voltage is supplied to the electrodes, allowing charge-transferring (also known as faradaic or redox) events to occur. KeywordsĮlectrolysis Cathode Electrical potential Battery Introduction Electrolytes include molten salts like sodium chloride. The electrolyte is typically a dissolved ion solution in water or other solvents. There are three pieces to an electrolytic cell: an electrolyte and two electrodes (a cathode and an anode). Electrolysis is a process that employs the utilisation of a Direct Electric Current (DC). An electrolytic cell is used to electroplate copper, silver, nickel, or chromium. Electrolysis is used to break down water into hydrogen and oxygen, as well as bauxite into aluminium and other compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
